A modern neurophysiological model of inhibition in tendinopathy: let’s move beyond ‘we need to reverse your cortical inhibition’
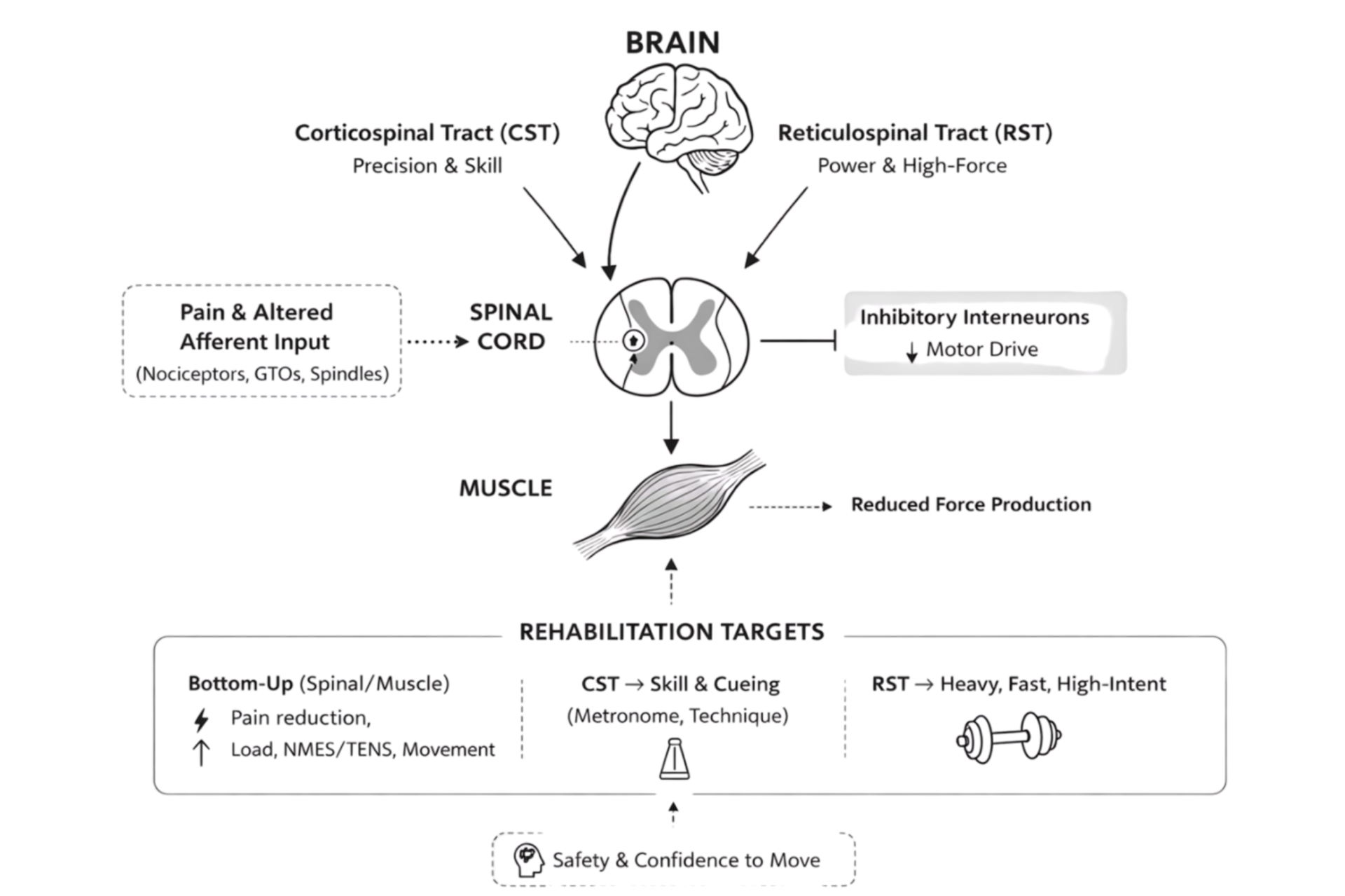
Spinal and muscle level reflexes
It is important to start peripherally, as this is where tendon pathology and pain originate. In a painful and pathological tendon there are nociceptors sensing there is an issue and relaying this to the spinal cord. This activates inhibitory interneurons located where nociceptors relay this signal, which then dampens concurrent descending spinal level motor drive, impacting the ability of the muscle to produce force [1]. Note though, nociceptive input is sufficient but not necessary to trigger this response. These same spinal reflexes can be activated via altered mechanoreceptor input (golgi tendon organs, muscle spindle) [1]. The nervous system at this spinal level is not sure what is going on given the barrage of different mechanical input from a pathological tendon, so it errs towards protection and dampens output.
What is described here is the tendon version of Arthrogenic
Muscle Inhibition or AMI. We provide evidence for this inhibition level in our
recent study [2]. What we showed is that activation of pain facilitation
circuits, which allow more signal to relay from nociceptors, was associated
with greater spinal level inhibition of the quadriceps in patellar
tendinopathy.
Cortical level problems
The prevailing thinking currently in tendinopathy is that
there is increased motor cortex inhibition, specifically corticospinal
inhibition, which might be responsible for altered motor output such as reduced
strength. This is based on a study by Rio et al. who studied athletes with
patellar tendinopathy [3].
While that study did identify there is altered motor drive between the brain
and the quadriceps, it was not able to pin-point where the inhibition was based
with the techniques used, and if this was relevant to altered function in
tendinopathy (corticospinal inhibition might be altered, but this does not
preclude spinal/muscle inhibition dominating the picture as peripheral changes
dominate over central changes) [4].
In other words, and to explain it simply, there is an interaction between
cortical levels and the spinal/muscle level. Motor output can be altered
by the central level (more inhibition within the motor cortex reduces the
signal descending towards the muscle) which may explain what the Rio study
found. However, the activity of the corticospinal tract is far less relevant if
inhibitory interneurons at the spinal/muscle level inhibit motor drive from
passing further downstream. It is also worth noting a different study did
actually measure the spinal/muscle level directly in people with patellar
tendinopathy, and they found it was inhibited [5].
However, that study didn’t assess the cortical level, so the full picture is
still not entirely clear.
Importantly, there are multiple within-brain level
connections (e.g. between the thalamus and motor cortex) and brain-spinal
connections (e.g. neural circuits that ‘modulate’ pain, such as those
responsible for a runner’s high, or pain relief with massage). As a result,
both central and spinal/muscle levels responsible for motor output can be influenced
by fear and catastrophising. So, for example, if someone feels safe to move,
the brain could send a signal to the spinal cord that is locked in an
inhibitory loop to say, don’t worry, we are safe, increase motor drive.
However, this is only likely to be partially successful unless the cause of the
local spinal/muscle level inhibition is addressed (more on that below).
The story of cortical inhibition is more complex than what we have been told
A critical but often overlooked point in the tendinopathy
literature is that the motor system uses two major descending pathways: the
corticospinal tract and the reticulospinal tract. The corticospinal tract is
specialised for precise, skill-based movements and plays a dominant role in
fine motor control, particularly in the upper limb. In contrast, the
reticulospinal tract is adapted for powerful, rapid, high-force movements
(think high intent, ‘violent’ movements) and contributes strongly to gross
motor output, postural control, and whole-limb actions [6].
This makes it particularly relevant for lower-limb tasks where the goal is to
rapidly generate force to move the body mass (for example, during sprinting or
jumping).
The reason the distinction between these tracts is important is that we have emerging evidence that the reticulospinal tract may be more influential when motor drive is dampened in lower limb tendinopathy. While there appears to be inhibition somewhere along corticospinal pathway at a spinal level, we now know that athletes with patellar tendinopathy increase the excitability of their reticulospinal tract to overcome this as, and this is the critical point, it can directly tune down inhibitory spinal reflexes [7]. This means it can ‘bypass’ inhibition if this is present in the corticospinal tract, restoring motor output. This is important when considering rehabilitation strategies (as discussed below), as interventions we can use to target motor drive for each tract differs [8, 9].

Strategies to address inhibition
A key point is we need to start at the spinal/muscle level because strategies to release central level inhibition are going to be much less effective if we don’t.
For the spinal level, we use bottom-up strategies.
This includes NMES, TENS, or any other strategy that helps pain (exercise, load
management, other local techniques or modalities).
For the central level, we can use top-down strategies
which vary depending on the tract that we are targeting. For the corticospinal
tract, we can use external cueing (e.g. metronome) and skill-based exercises
(from calf raises to hops and everything in between, if there is a
skill/learning element). For the reticulospinal
pathway, we use hard and fast (in combination) and intent-driven, high-effort
tasks (heavy load calf raises with max CONC intent are an example) [7, 8].
In tendinopathy what has become popular is metronome use during calf raises, stair climbing and oscillating calf raises. The issue here is they mainly target the corticospinal pathway, which may not lead to the best outcome in isolation.
So, what should we do in practice?
The best option is to: a/ target pain as a priority; and b/
try and normalise movement to improve afferent mechanoreceptor firing to target
the spinal/muscle level issues as a priority. Also move onto 1/ Skill and metronome-based
tasks; but importantly also 2/ heavy/fast intent-based loading to ensure you
are addressing potential central level issues via both descending tracts.
Probably most critical of all is the head space the person is in. If they are not feeling safe to move, this will have a massive top-down influence on their motor output, regardless of the dominant neurophysiological sources of inhibition. Higher brain centres interact with motor drive at central and spinal levels and can have a powerful inhibitory influence. So our number one priority is the ensure the person feels safe via sense making and movement-mediated embodied self-efficacy.
So, there we have it! We hope this was useful.
Peter Malliaras and Patrick Vallance
References
1. Lepley, A.S. and L.K. Lepley, Mechanisms of arthrogenic muscle inhibition. Journal of sport rehabilitation, 2021. 31(6): p. 707-716.
2. Vallance, P., Kidgell, D.J. and Malliaras, P., Greater endogenous pain facilitation is associated with lower spinal excitability and maximal knee extension strength deficits in athletes with patellar tendinopathy. European journal of applied physiology, 2026. Feb 25. doi: 10.1007/s00421-026-06166-0. Online ahead of print.
3. Rio, E., et al., Elevated corticospinal excitability in patellar tendinopathy compared with other anterior knee pain or no pain. Scandinavian Journal of Medicine and Science in Sports, 2016. 26(9): p. 1072-1079.
4. Vallance, P., et al., Transcranial magnetic stimulation and electrical stimulation techniques used to measure the excitability of distinct neuronal populations that influence motor output in people with persistent musculoskeletal conditions: A scoping review and narrative synthesis of evidence. Journal of Electromyography and Kinesiology, 2025: p. 103011.
5. Davi, S.M., et al., Quadriceps inhibition after naturally occurring patellar tendon damage and pain. Journal of athletic training, 2020. 55(6): p. 608-614.
6. Akalu, Y., et al., Identifying the role of the reticulospinal tract for strength and motor recovery: A scoping review of nonhuman and human studies. Physiological Reports, 2023. 11(14): p. e15765.
7. Vallance, P., P. Malliaras, and D.J. Kidgell, Temporal motor evoked potential decomposition reveals that cortico-reticulospinal excitability is increased in athletes with patellar tendinopathy – preliminary evidence of compensatory adaptation for diminished motor drive. Under review.
8. Lecce, E., et al., Resistance training‐induced adaptations in the neuromuscular system: Physiological mechanisms and implications for human performance. The Journal of Physiology, 2026. 604(1): p. 81-115.
9. Akalu, Y., et al., Determining the cortical, corticospinal, and reticulospinal responses to metronome-paced and self-paced strength training. European Journal of Applied Physiology, 2025: p. 1-24.

Connect with Prof Peter & the Team
Book a consultation
-
Visit Peter at our Richmond Clinic
-
Via a Telehealth session
-
132 Bridge Road, Richmond 3121, Melbourne Australia
